CONTACT
Department of Chemistry
Todd Wehr Chemistry, 101
1414 W Clybourn St
PO Box 1881
Milwaukee, WI 53233
(414) 288-3515
Problem with this webpage?
Report an accessibility problem
Any other problem, contact muchem@marquette.edu.
 Dr. James R. GardinierMarquette University
Dr. James R. GardinierMarquette UniversityWehr Chemistry Building, 438
MilwaukeeWI53201United States of America(414) 288-3533james.gardinier@marquette.eduPh.D., University of Buffalo
B.S., University of Buffalo
My group's research is in the general area of inorganic supramolecular and materials chemistry. We are interested in a variety of topics with an emphasis on studying ways to manipulate electron or energy flow in supramolecular assemblies with the purpose of designing new molecular wires, fluorescent dyes, and/or light harvesting antennae. A second emphasis is on studying the role of noncovalent interactions on the assembly of molecules in the solid state and on what impact crystal packing behavior has on the bulk properties of matter. The multidisciplinary nature of inorganic supramolecular chemistry affords the opportunity for undergraduate, graduate students and postdoctoral researchers in my group to become acclimated to a variety of synthetic techniques spanning the traditional fields of organic, organometallic, and inorganic chemistry. Each research project also offers opportunities to learn fundamental skills in the areas of magnetism, electrochemistry, photochemistry and spectroscopy, as well as X-ray crystallography. The research projects briefly mentioned below are more fully described in my group's home page.
Tunable Fluorescent Dyes
We have recently developed new classes of compounds based on 2-pyrazolylaniline (pzAn) derivatives that can act as fluorescent sensors for a variety of Lewis acids. Some advantages that these new compounds enjoy over their competition are simple, high yield (nearly quantitative) syntheses, the ability to tune the wavelength of fluorescence by very simple modifications of the ligand backbone, and a selective fluorescence response depending on the nature of the Lewis acid. The fluorescence tunability is exemplified by the picture on the right, which shows a series of diphenylboron complexes of different pzAn derivatives irradiated with 354 nm UV light. The ultimate goals of this research project include using these derivatives as sensitizers for enhancing lanthanide-based emissions, using these fluorescent dyes as biological markers for studies in drug metabolism studies, and incorporating these fluorophores onto semiconductor surfaces to serve as light harvesting antenna in solar cell applications.

Molecular Wires
Designing components for future use in molecular electronics is another area of interest to our research group. Towards this end, we are investigating the preparation, electrochemistry, and self-assembly capabilities of metal chain compounds, pi-stacked N-heterocycles, and/or combinations thereof as model wire systems. The ultimate goal of this research is to firmly establish the geometric limits and possible metal/heterocycle substitution patterns necessary for efficient intramolecular charge propagation. A primary challenge in this research is to expand the currently known ligand systems that can be used to form metal chain compounds, where neighboring metals are disposed within van der Waals contact. Specifically, it would be desirable to examine discrete systems with increasing number of interacting electroactive centers in order to establish possible trends in the electrochemical or even spectroscopic data. On paper, this would seem to be a simple order to fulfill, however, when dealing with multinucleating ligands one always has to be concerned about the possibility for supramolecular isomerism. This is exemplified in the simplest case scenario in the silver coordination chemistry of some of our dipyrazolyl-based systems in Figure 1. Our group actively investigates the factors that will lead to the preferential formation of supramolecular isomers in these and more complex derivatives. We also examine the differences in the electrochemical and spectroscopic behaviors of each type of supramolecular isomer to gain knowledge in to how to design better systems. Another set of potential wires we look at are based on Jekyll-and-Hyde scorpionates exemplified by the alkali metal salt shown in Figure 2. With these electroactive dipolar ligands, charge can propagate via the pi system of the ligands. We construct one-dimensional chains based on Pearson’s Hard soft acid-base theory and examine their electrochemical and spectroscopic properties, to evaluate their potential in future wire systems.
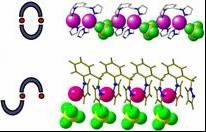
Figure 1
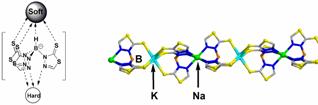
Figure 2
Spin Crossover Compounds
Transition metal complexes with d4-d7 electron configurations have the capability of exhibiting two spin states (high spin or low spin) depending on the strength of the ligand field. Strong field ligands tend to favor electron pairing, giving low spin complexes while weak field ligands give rise to high spin complexes with the maximum number of unpaired electrons. Spin crossover (SCO) compounds are special cases where the transition metal complexes have ligands or ligand combinations of intermediate field strength. In such cases, the high or low spin states are in equilibrium and it is possible to access either the high or low spin state depending on external perturbations such as light, heat, or pressure. The change in spin state is accompanied by changes in magnetic moment, color and molecular geometry. The picture to the right shows an example of the thermochromic behavior of an iron(II) SCO compound on warming from 90K to 273K (left to right); it is easy to see why researchers have been fascinated by this sort of compound since their discovery in the 1960’s. The reversible change in magnetic moment and color upon heating, on application of pressure, or upon laser stimulation, holds promise for the use of such materials in different sensing applications. In order to realize such applications several issues need to be addressed, which are the focal points of our and of other’s research interest around the world. First, can we learn to control the spin crossover behavior (temperature/pressure range) for custom designing future thermochromic or pressure-sensing materials? Secondly, how can we make these SCO materials robust enough for practical sensing applications? Often, but not always, the SCO compounds are susceptible to crystal fracture (via desolvation or stress) which can subsequently change the SCO properties of the material. An example of such a decomposition is shown on the right for a different iron(II) SCO compound going through one heating and cooling cycle; we can show that the magnetic properties of the virgin material are different than the aged sample in many cases- this is clearly not very desirable for incorporation into a reliable device. One example of a strategy we have used is to make robust and potentially useful SCO materials is to incorporate them into polymer matrices, as in Figure 3.


Figure 3
Professor Gardinier received his Ph.D. degree in Inorganic chemistry under the direction of Ted Beachley from the University at Buffalo in 2000. After completing post-doctoral training in the general area of Inorganic Supramolecular Chemistry with François Gabbaï at Texas A&M (1999-2001) and with Dan Reger at the University of South Carolina (2001-2004), he joined the faculty at Marquette University in August 2004. Professor Gardinier’s research interests are in the area of inorganic supramolecular chemistry/materials chemistry.
Professor Gardinier is a recipient of the 2008 Way Klingler Young Scholar Award.