CONTACT
Department of Chemistry
Todd Wehr Chemistry, 101
1414 W Clybourn St
PO Box 1881
Milwaukee, WI 53233
(414) 288-3515
Problem with this webpage?
Report an accessibility problem
Any other problem, contact muchem@marquette.edu.
 Dr. Chae S. YiMarquette University
Dr. Chae S. YiMarquette UniversityWehr Chemistry Building, 619
MilwaukeeWI53201United States of America(414) 288-3536chae.yi@marquette.eduThe Yi LabPh.D., University of Wisconsin-Madison
B.S., University Michigan-Ann Arbor
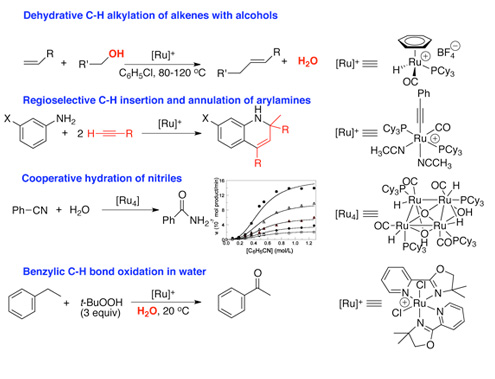
Transition metal catalyzed C-H bond activation reaction is a powerful synthetic method for forming functionalized products directly from unreactive hydrocarbons, and has enormous synthetic potentials for developing chemical processes ranging from petroleum products to pharmaceutical agents. Catalytic C-H bond activation method also has high potential for “reforming” fossil fuels and biomass for obtaining clean and environmentally sustainable chemical energy. One of the long term goals of our group’s research to design effective metal catalysts, which can facilitate efficient and selective C-H bond activation reactions of synthetic importance. This research group recently discovered a number of highly effective coupling reactions involving C-H bond activation (see below reaction scheme) by using well-defined ruthenium catalysts. We are currently investigating the detailed reaction mechanisms of these catalytic reactions, and are searching for novel C-H coupling reactions of synthetic importance by using well-defined ruthenium catalysts.
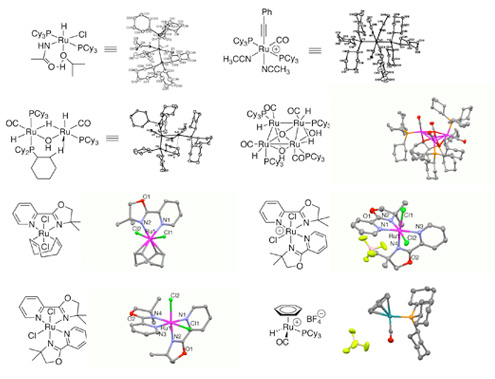
Another major area of our group’s research centers on the synthetic and mechanistic study of the cooperative reactivity of multinuclear ruthenium catalysts for synthetically useful chemical transformations. Cooperative catalysis by multinuclear transition metal complexes has long been regarded as an effective way to control and regulate chemical reactions that are normally difficult to achieve from monometallic ones, and can serve as functional models for natural allosteric metalloenzymes. Multinuclear ruthenium cluster complexes have long been considered as functional models for promoting cooperative activity for unreactive C-H, C-C and C-N bond activation reactions. We recently discovered that the bi- and tetrametallic ruthenium-hydroxo complexes are highly cooperative catalysts for the alcohol oxidation and nitride hydration reactions, and are studying to attain fundamental mechanistic knowledge on the cooperative nature of the ruthenium-hydroxo catalysts for these reactions. We are also engaged in designing and utilization of new cooperative metal catalysts for other coupling reactions involving C-H bond activation.
Selected examples of the catalytic reactions developed in our research group:
GROUP MEMBERS
The group is composed of three graduate students:
· Krishna Gnyawali
· Aldiyar Shakenov
· Dulanjali Thennakoon
Professor Yi received his B.S. degree from the University Michigan-Ann Arbor in 1987 and Ph.D. from the University of Wisconsin-Madison in 1991. After completing a postdoctoral training at the Pennsylvania State University (1992-93). He joined the department in 1993, was promoted to Associate Professor with tenure in 2000 and Professor in 2011. His area of research is inorganic chemistry, and has been teaching undergraduate/graduate courses in that area. His publication record includes over 46 publications in inorganic chemistry, most of them in American Chemical Society journals. He recently received an NSF grant studying catalytic C-H bond activation reactions. This work will develop new "green" catalytic processes that will give desired products efficiently without forming wasteful byproducts under environmentally compatible conditions.
Please see the full list on Google Scholar.