Emeriti & Former Faculty
CONTACT
Department of Biological Sciences
Wehr Life Sciences, 109
1428 W. Clybourn St.
Milwaukee, WI 53233
(414) 288-7355
 Dr. Edward BlumenthalMarquette University
Dr. Edward BlumenthalMarquette UniversityWehr Life Sciences, 504
MilwaukeeWI53201United States of America(414) 288-6270edward.blumenthal@marquette.edue-PublicationsB.A. (Biochemistry), 1987, Cornell University, Ithaca, NY
Ph.D. (Neuroscience), 1993, Yale University, New Haven, CT
Postdoctoral Fellow, University of California-San Diego, CA
Postdoctoral Fellow, University of Virginia, Charlottesville, VA
BIOL 1001 Introductory Biology 1
BIOL 2953 Entering Research
BIOL 3702 Experimental Physiology
BIOL 4201 Genomics and Bioinformatics
BIOL 6005 Scientific Writing
BIOL 8704 Cellular Homeostasis
My laboratory focuses on two different aspects of the biology of the fruit fly Drosophila melanogaster. With both projects, are goal is combine multiple experimental approaches--genetic, molecular, and physiological/behavioral--to understand as fully as possible the processes that we are studying. Descriptions of the two projects are given below.
We are studying the adult lethal gene drop-dead (drd) which, when mutated, causes flies to die during the first week of adult life. The drd gene encodes a so-called NRF domain protein—a member of a family of integral membrane proteins with some homology to bacterial acyl transferases. No biochemical function has yet been determined for any eukaryotic NRF domain protein. While drd has previously been reported to cause neurodegeneration, we have found that the mutant flies also have many other phenotypes. First, they have a problem moving food through their guts, as they end up with a large amount of food stuck in the crop, which is a food storage organ (see figure below). Second, mutant female flies are sterile, due to a defect in the cross-linking of some of the proteins that make up the eggshell.
In our lab, we make use of a variety of different genetic and biochemical techniques to understand the function of drop-dead. Recently, we have found that drd expression is required during metamorphosis, but not during adulthood, for adult viability. Also, we find that expression in multiple tissues is required for survival, suggesting that there might be multiple “causes of death” when the drd mutant flies die. Ongoing projects in the lab include screening for genes that lie in the same metabolic pathway as drd, identifying the drd expression pattern, and studying the defect in eggshell formation in drd mutant females.
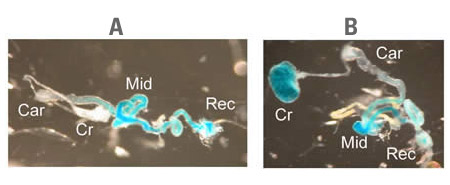
Distribution of food within the gut of wild-type and mutant flies. On the left is the dissected gut of a wild-type fly that was kept on blue food. Note that food is present in the midgut (Mid) and rectum (Rec) but not in the crop (Cr) or cardia (Car). In contrast, the gut from a drd mutant fly (right) has a large amount of food in the crop and some staining also in the cardia.
Many of the important homeostatic processes that are necessary for life, such as regulation of water and ionic balance or the absorption of nutrients, are mediated by transport across epithelial cell layers. Precise control of such transport pathways is essential for an organism to adapt to a changing environment. In this project we are studying a model transport epithelium, the Malpighian tubules, which function as the fly’s kidney. The Malpighian tubules produce urine by transporting ions—primarily potassium and chloride—and water out of the hemolymph and into the tubule lumen, which is contiguous with the gut. The rate of urine secretion is controlled by many diuretic and antidiuretic factors in response to changes in the hydration and feeding state of the fly. We have discovered that the biogenic amine tyramine (which is the compound found is red wine and cheese that some have linked to migraines) acts as one of these diuretic factors (Blumenthal, 2003). Application of tyramine causes an increase in the conductance of chloride across the tubule and thus an increase in urine production. We also find that not only can the tubule respond to tyramine, it can also synthesize it from the amino acid tyrosine. As shown in the figure below, we have found that tyramine synthesis and reception occur in different cells in the tubule. Tyramine is synthesized in the principal cells from tyrosine by the enzyme tyrosine decarboxylase, encoded by the gene Tdc1. The tyramine then activates receptors on the stellate cells—primarily the receptor encoded by the gene tarot (tro, for TA Receptor Of the Tubule, aka CG7431). This is the first example of cell-cell communication in an insect Malpighian tubule. Current projects ongoing in the lab involve characterizing second messenger pathways that modulate the response of the tubule to tyramine and identifying other gene products necessary for the synthesis and release of tyramine.
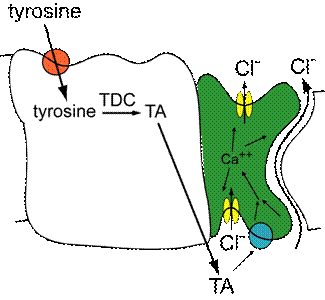 Model of tyramine (TA) action in the Malpighian tubule. In our model, tyrosine is taken up into the principal cells of the tubule (white) and converted into tyramine by the enzyme tyrosine decarboxylase (TDC). The tyramine is released from the principal cells and binds to receptors on the stellate cells (green), stimulating an increase in chloride conductance and urine secretion.
Model of tyramine (TA) action in the Malpighian tubule. In our model, tyrosine is taken up into the principal cells of the tubule (white) and converted into tyramine by the enzyme tyrosine decarboxylase (TDC). The tyramine is released from the principal cells and binds to receptors on the stellate cells (green), stimulating an increase in chloride conductance and urine secretion.
Genetics Society of America
The American Physiological Society
Society for Experimental Biology
Entomological Society of America
Conway, S., Sansone, C.L., Benske, A., Kentala, K., Billen, J., Vanden Broeck, J., and Blumenthal, E.M. 2018. Pleiotropic and novel phenotypes in the Drosophila gut caused by mutation of drop-dead. Journal of Insect Physiology 105: 76-84. doi: 10.1016/j.jinsphys.2018.01.007
Zhang, H. and E.M. Blumenthal. 2017. Identification of multiple functional receptors for tyramine on an insect secretory epithelium. Scientific Reports 7(1):168. doi: 10.1038/s41598-017-00120-z.
Sansone, C.L. and E.M. Blumenthal. 2013. Neurodegeneration in drop-dead mutant Drosophila melanogaster is associated with the respiratory system but not with hypoxia. PLoS One, 8.7:e68032. doi:10.1371/journal.pone.0068032.
Sansone, C.L. and E.M. Blumenthal. 2012. Developmental expression of drop-dead is required for early adult survival and normal body mass in Drosophila melanogaster. Insect Biochemistry and Molecular Biology, 42.9:690-8. doi:10.1016/j.ibmb.2012.06.002.
2011-12 Senior Research Fellowship, KU Leuven, Leuven Belgium
2014-2019 Regulation of epithelial barrier formation in Drosophila. NSD IOS-1355087
2012-2015 REU Site: Undergraduate Summer Research in Molecular and Cellular Biology. NSF DBI-1156569
2008-2013 Characterization of tyramine-mediated cell-cell communication in the Drosophila Malpighian tubule. NSF IOS-0744619
2007-2010 Characterization of a mutation that affects feeding and digestion in Drosophila. NIGMS R15-GM080682
2001-2003 Identification of osmoregulatory mutants in Drosophila. NIDDK R21-DK080680
Regulation of extracellular barrier integrity by the drop-dead gene in Drosophila melanogaster. 1st International Molecular Plant Protection Congress. Adana, Turkey. April 10-13, 2019. (Invited keynote speaker).
Regulation of larval growth by drop-dead. 60th Annual Drosophila Research Conference. Dallas, TX. March 27-31, 2019. (Presented by L. Hopson)
Identifying pathways responsible for epithelial barrier formation in Drosophila. Department of Biological Sciences, Marquette University, Milwaukee, WI. May 19, 2017.
drop-dead and the formation of extracellular barriers in Drosophila. Department of Biochemistry and Molecular Biophysics, Kansas State University, Manhattan, KS. November 29, 2017.
Two cytochrome p450s essential for peritrophic matrix synthesis. 58th Annual Drosophila Research Conference. San Diego. March, 2017. (Presented by S. Conway).
Identification of lipid-processing genes required for eggshell formation in Drosophila. 58th Annual Drosophila Research Conference. San Diego. March, 2017. (Presented by K. Gackowski). 3rd Place Winner, Undergraduate Poster Award.
Role of drop-dead in spermatogenesis. 58th Annual Drosophila Research Conference. San Diego. March, 2017. (Presented by A. Benske).
Knockdown of multicopper oxidase 4 eliminates the peritrophic matrix and alters larval growth and adult microbiome in Drosophila. SEB Annual Meeting. Gothenberg, Sweden. July 5, 2017. (Presented by E.M. Blumenthal).
Life without a peritrophic matrix: effect of eliminating the PM in Drosophila melanogaster. Entomology 2017. Denver, CO. November 7, 2017. (Presented by E.M. Blumenthal) (invited talk).
Knockdown of multicopper oxidase 4 eliminates the peritrophic matrix and alters the adult microbiome in Drosophila. Midwest Drosophila Conference. Monticello, IL. November 5, 2017. (Presented by S. Conway). 2nd Place Winner, Best Student Talk.
Examining the role of drop-dead in spermatogenesis. Midwest Drosophila Conference. Monticello, IL. November, 2017. (Presented by A. Benske).
Dr. Blumenthal is currently accepting new Ph.D. students into his lab
Unmila Purkaystha Jhuti (PhD student)
Nate Fischer (undergraduate)
Dina Kaloti (undergraduate)
Kyle Yu (undergraduate)
Oliwia Burda (undergraduate)
Julie Bucheger (2004, B.S.)
Joe LaPlaca (2004, B.S. )
Jen Krueger (2005, B.S.)
Graham Smith (2005, B.S.)
Tiffani Cherry (2006, B.S.)
Amanda Herman (2006, B.S.)
Matt Klinker (2004, B.S.)
Will Mueller (2007, B.S.)
Cassie Peller Nelson (2008, B.S.)
Nic Haas (2008, B.S.)
Elizabeth Bacon (Summer Research Program, 2007)
Laura Korthauer (2009, B.S.)
Jackie Whelan (2009, B.S.)
Olivia Corradin (2010, B.S.)
Jennifer Rorex (2010, B.S.)
Kristen Ruka (2010, B.S.)
Kendall Knight (Summer Research Program, 2010)
Francisco Nava (2011, B.S.
Megan Mohnen (2013, B.S.)
Anna Quint (2012 B.S.)
Katie Schober (2012 B.S.)
Tayler Sheahan (2012 B.S.)
Sam Schultz (undergraduate technician)
Catharine Skoog (undergraduate researcher)
Dr. Haiying Zhang (postdoctoral fellow)
Chrissy Sansone (2013, Ph.D.)