Emeriti & Former Faculty
CONTACT
Department of Biological Sciences
Wehr Life Sciences, 109
1428 W. Clybourn St.
Milwaukee, WI 53233
(414) 288-7355
 Dr. Emily M. SontagMarquette University
Dr. Emily M. SontagMarquette UniversityWehr Life Sciences 209
MilwaukeeWI53201United States of America(414) 288-7792emily.sontag@marquette.edue-PublicationsB.S., 2005, Rose-Hulman Institute of Technology, Terre Haute, IN
Ph.D., 2011, University of California, Irvine
Post-doctoral Fellow, 2013, Stanford University, Stanford, CA
The Sontag Lab utilizes microscopy and biochemistry techniques to understand the cellular and molecular mechanisms underlying cellular stress responses to misfolded proteins and their role in disease. Neurodegenerative diseases (such as Alzheimer’s, Parkinson’s, and Huntington’s diseases), cancer, and even aging are all linked to protein misfolding. A major goal of the lab is to better understand how the cell responds to misfolded proteins, so that we can learn what goes wrong during disease and develop new therapeutic strategies to treat these disorders.
We are currently focused on studying neurodegenerative diseases. A common aspect of these disorders is misfolded proteins accumulating into large deposits within the brain. These deposits are formed when misfolded proteins are sequestered into membrane-less organelles or compartments.
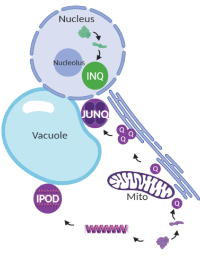 The function of these compartments is unknown, but an intriguing hypothesis is that they prevent aberrant protein interactions and facilitate clearance of protein aggregates by alternative pathways when the protein clearance machinery cannot keep up with the misfolded protein load. Misfolded protein sequestration into distinct compartments is conserved from yeast to mammals, indicating this mechanism of protein quality control is of critical importance for cell health and survival. We are currently utilizing yeast as a model system to understand the basic cell biology of protein quality control.
The function of these compartments is unknown, but an intriguing hypothesis is that they prevent aberrant protein interactions and facilitate clearance of protein aggregates by alternative pathways when the protein clearance machinery cannot keep up with the misfolded protein load. Misfolded protein sequestration into distinct compartments is conserved from yeast to mammals, indicating this mechanism of protein quality control is of critical importance for cell health and survival. We are currently utilizing yeast as a model system to understand the basic cell biology of protein quality control.
Figure 1. Spatial protein quality control compartments in yeast.
We are working to answer 2 major questions:
We are looking for motivated students from all races, ethnicities, genders, and socio-economic status to join us in answering these questions and more!
Sarah Rolli (Ph.D. student)
Chloe Langridge (Ph.D. student)
Dr. Sontag is currently accepting new Ph.D. students into her lab