Tayebi Research Group Current Research
We envision a future where medical treatment has become highly personalized, with treatment modalities that are patient specific, not ‘one fits all’. Such a future requires applications that possess high performance, embedded biosensors into feedback-based devices, smart biomaterials and advanced diagnostics. Such functionality will necessitate a technology with increased sensitivity, specificity, tunability, and that is customizable. While this technology has yet to be fully realized, recent advances in biofabrication, bioplastics and bioprinting (microcontact printing, inkjet printing, and three-dimensional (3D) printing) may enable on-demand and patient-specific medical treatments.
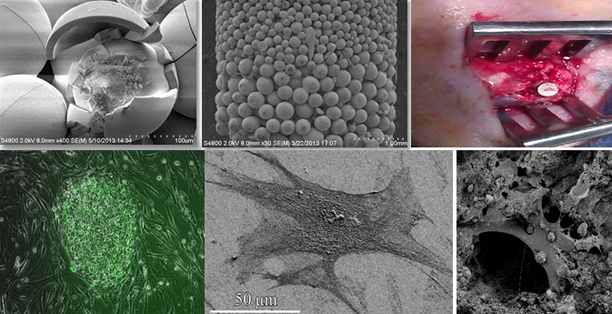
Current Research Projects
Hard and Soft Tissue Expansion, Vascularization and Stem Cell Seeding in Patient Specific 3-D Printed Scaffold for Large Craniofacial Defects
Acute or refractory craniofacial defects and disorders present surgeons with numerous reconstructive challenges. For example, conditions such as oral cancer, locally aggressive benign tumors (such as ameloblastoma), jaw cysts, osteomyelitis, gunshot wounds, osteoradionecrosis, and medication-related osteonecrosis of the jaw (MRONJ) require extensive surgical resection and reconstruction of the affected area. Smaller defects, such as those seen in dentoalveolar ridges after trauma, periodontal disease, and partial anodontia cases also require challenging surgical reconstruction methods.
Such defects are currently reconstructed using microvascular free flaps, non-vascular autogenous bone and/or skin grafts, or reconstruction with xenografts or synthetic materials. Using these methods, surgeons are constrained by the size and complexity of the defect; defects larger than 5 cm are difficult to reconstruct with these techniques, so are those defects that involve the anterior mandibular region. Lack of predictability of graft success, as well as the lack of predictability of the esthetic and functional outcome of a non-vascular grafting makes it necessary to use a vascularized free flap in many of these cases. Such procedures are lengthy and challenging despite the improvements in the technology and the techniques. Patients experience long recovery periods and are exposed to related perioperative morbidities that increase health care costs, increase time lost from work, and negatively affect the patients’ quality of life. 3D printing is currently being intensely studied by a diverse set of applications, including the development of bioengineered tissues, as well as the production of functional biomedical materials and devices for dental and orthopedic applications. We apply 3D printing technology to develop solutions that address the issues of cost and customization through designs that are patient-specific and conformable to the patients’ unique needs. Our primary focus is the creation of novel bioactive scaffolds for in situ tissue repair and generation. These scaffolds will be tailored to meet the patient’s current condition, age, and prior medical history and designed for tension free soft tissue closure, enhanced vascularity and tissue formation, and the prevention of micromobility.
Tissue Engineering and Regenerative Implants: A 3D-BioPrinting Approach
The main challenge in tissue engineering is designing appropriate scaffolds with suitable material properties to repair, replace or regenerate damaged tissues. Ongoing investigation in our laboratory is focused on improving the mechanical and electrical characteristics of the scaffolds with respect to their biocompatibility, bioactivity and biodegradability along with their in vitro and in vivo studies.
Moreover, we are focusing on the biology, biomechanics, design and surface modification of structures for artificial materials, polymer/metal implants, bioactive ceramics, and delivery systems to be installed in surgical implants. We analyzed the biocompatibility and detailed performance analysis of our systems by in vitro characterizations using stem cells.

Microfluidic Systems for Medical Applications
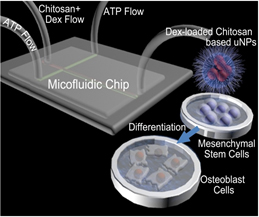
Stem-cell therapies have the potential to revolutionize healthcare. In this context, applying bioactive agents such as growth factors and drugs for controlling the in situdifferentiation of stem cells is indispensable. Although a variety of vehicles have been investigated for the delivering of bioactive agents, a reliable, cost-effective and robust method has yet to be developed. More specifically, current methods for fabrication of micro-/nanoparticle (μNP) vehicles, such as the conventional bulk mixing method, result in extremely polydisperse μNPs that exhibit various physicochemical properties which are difficult to control and not appropriate for scale-up manufacturing processes. These methods often require additional costly and time-consuming post-processing steps.
We have recently reported on a transformative innovation in biomanufacturing of cargo-loaded μNPs using a solvent-free microfluidic technique and a carrier-based intracellular delivery system. Our bottom-to-top approach has a major advantage of control over the properties of μNPs through adjustment of flow parameters.
We use this microfluidics platform and examine sustained release of both small and large sized molecules in customized μNPs. We expect that our efforts will result in a universal and robust protocol for the encapsulation of a diverse set of drugs and growth factors that regulate stem cell differentiation. We will also develop a theoretical understanding of cellular interactions and level of polymer/cell membrane connections by employing first principle quantum mechanics (QM) and molecular dynamics (MD) simulation tools.
A Novel Approach in Teeth Preservation
A frequent scenario that happens for an endodontist: a mother runs to the doctor’s office with a tooth of her child in hand. The child has lost a tooth due to a trauma in a playground, causing complete displacement of the tooth from its socket in alveolar bone. The mother asks for the tooth to be returned into the mouth. In many cases the tooth is no longer good. The tooth could have been saved if the mother had used a right storage media.
Adult teeth are avulsed from the mouth frequently in adolescence. Successful replacement of the tooth into the mouth is limited by the type of storage media available. Ideal storage media is expensive and expires quickly. If a tooth is out of the socket for more than 30 minutes the tooth most likely will be lost within 5 years. This study will attempt to preserve periodontal ligament cells and identify a storage medium that is inexpensive, readily available and will preserve the avulsed tooth for longer than 30 minutes.
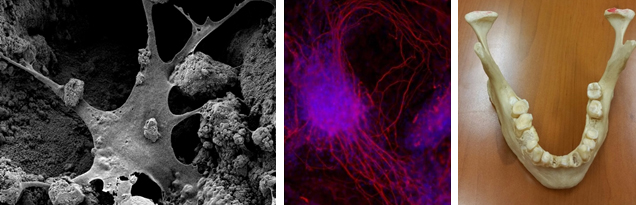
Regenerative Medicine Treatment of Large Bone Defect
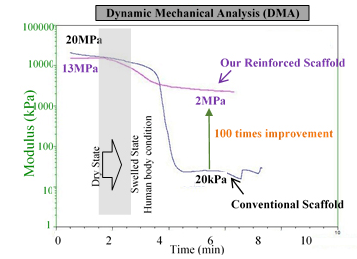 Metallic implants are widely used in bone treatment, but unfortunately these implants are not degradable, and second surgery is often required for their removal from the body. There have been serious attempts among researchers and manufacturers to produce biodegradable implants/scaffolds to avoid the need for the second surgery. However, poor mechanical strength in these products prohibits their commercialization.
Metallic implants are widely used in bone treatment, but unfortunately these implants are not degradable, and second surgery is often required for their removal from the body. There have been serious attempts among researchers and manufacturers to produce biodegradable implants/scaffolds to avoid the need for the second surgery. However, poor mechanical strength in these products prohibits their commercialization.
In this proposal, we are proposing a hybrid biodegradable scaffold with mechanical properties in the range of the human bone. The materials we are using in this scaffold are all FDA-approved and ready to go through the commercialization process. Our primary results verify 100 times enhancement in the mechanical strength of our new design scaffolds compared to the conventional ones.
Marquette University School of Dentistry, Lab S021,
1801 W. Wisconsin Ave, Milwaukee, WI 53201-1881
Copyright © Lobat Tayebi
All rights reserved




 Metallic implants are widely used in bone treatment, but unfortunately these implants are not degradable, and second surgery is often required for their removal from the body. There have been serious attempts among researchers and manufacturers to produce biodegradable implants/scaffolds to avoid the need for the second surgery. However, poor mechanical strength in these products prohibits their commercialization.
Metallic implants are widely used in bone treatment, but unfortunately these implants are not degradable, and second surgery is often required for their removal from the body. There have been serious attempts among researchers and manufacturers to produce biodegradable implants/scaffolds to avoid the need for the second surgery. However, poor mechanical strength in these products prohibits their commercialization.